Hypersomnia Therapeutics Market Size, Trends, and Growth Strategies 2025-2032
The Hypersomnia Therapeutics industry is witnessing a robust evolution driven by increasing prevalence of sleep disorders and advancements in treatment modalities. As emerging biopharmaceutical innovations reshape treatment landscapes, the market dynamics reveal significant potential for strategic investments and product development, aligning with shifting patient needs and regulatory frameworks.
Market Size and Overview
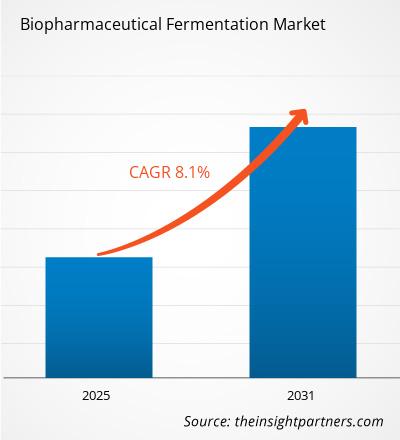
The Global Hypersomnia Therapeutics Market is estimated to be valued at USD 349.9 Mn in 2025 and is expected to reach USD 635.5 Mn by 2032, growing at a compound annual growth rate (CAGR) of 8.9% from 2025 to 2032.
Hypersomnia Therapeutics Market-https://www.coherentmi.com/industry-reports/hypersomnia-therapeutics-market
The Hypersomnia Therapeutics industry is witnessing a robust evolution driven by increasing prevalence of sleep disorders and advancements in treatment modalities. As emerging biopharmaceutical innovations reshape treatment landscapes, the market dynamics reveal significant potential for strategic investments and product development, aligning with shifting patient needs and regulatory frameworks.
Market Size and Overview
The Global Hypersomnia Therapeutics Market is estimated to be valued at USD 349.9 Mn in 2025 and is expected to reach USD 635.5 Mn by 2032, growing at a compound annual growth rate (CAGR) of 8.9% from 2025 to 2032.
Hypersomnia Therapeutics Market-https://www.coherentmi.com/industry-reports/hypersomnia-therapeutics-market
Hypersomnia Therapeutics Market Size, Trends, and Growth Strategies 2025-2032
The Hypersomnia Therapeutics industry is witnessing a robust evolution driven by increasing prevalence of sleep disorders and advancements in treatment modalities. As emerging biopharmaceutical innovations reshape treatment landscapes, the market dynamics reveal significant potential for strategic investments and product development, aligning with shifting patient needs and regulatory frameworks.
Market Size and Overview
The Global Hypersomnia Therapeutics Market is estimated to be valued at USD 349.9 Mn in 2025 and is expected to reach USD 635.5 Mn by 2032, growing at a compound annual growth rate (CAGR) of 8.9% from 2025 to 2032.
Hypersomnia Therapeutics Market-https://www.coherentmi.com/industry-reports/hypersomnia-therapeutics-market
0 Yorumlar
0 hisse senetleri
672 Views
0 önizleme